| Chemistry reagent discs are circular, injection molded diagnostic consumables containing multiple reagent chambers, microfluidic channels, and optical windows in a single integrated component. They are manufactured by plastic injection molding because the process achieves the ±0.01 mm dimensional tolerances required for precise fluid control, replicates complex three-dimensional channel geometries consistently across millions of units, and produces optically clear components with surface quality suitable for photometric and fluorescence-based detection. |
The accuracy of a diagnostic test — whether it measures blood glucose, liver enzymes, or electrolytes — depends as much on the precision of the disc that contains the reagents as on the chemistry itself. A reagent well with the wrong volume changes the reagent-to-sample ratio. A microfluidic channel with the wrong width alters the flow rate. A scratched optical window adds noise to the absorbance measurement.
Injection molding is the only manufacturing process capable of producing these complex, optically precise, multi-feature components at the volumes diagnostic testing demands — tens of millions of discs annually for a single analyzer platform. Let’s explore more.
What Are Chemistry Reagent Discs?
Chemistry reagent discs are precision-molded consumable components used in automated clinical chemistry analyzers and point-of-care testing (POCT) devices. Each disc integrates multiple pre-loaded reagent chambers, microfluidic transfer channels, and photometric measurement cuvettes into a compact circular form — typically 50–150 mm in diameter — designed to be loaded into a rotating analyzer platform.
The disc format is not arbitrary. Circular geometry allows the analyzer to use centrifugal force to precisely meter sample and reagent volumes into reaction cuvettes, mix them under controlled conditions, and position measurement cuvettes in front of optical detectors — all without pumps, external valves, or manual pipetting steps.
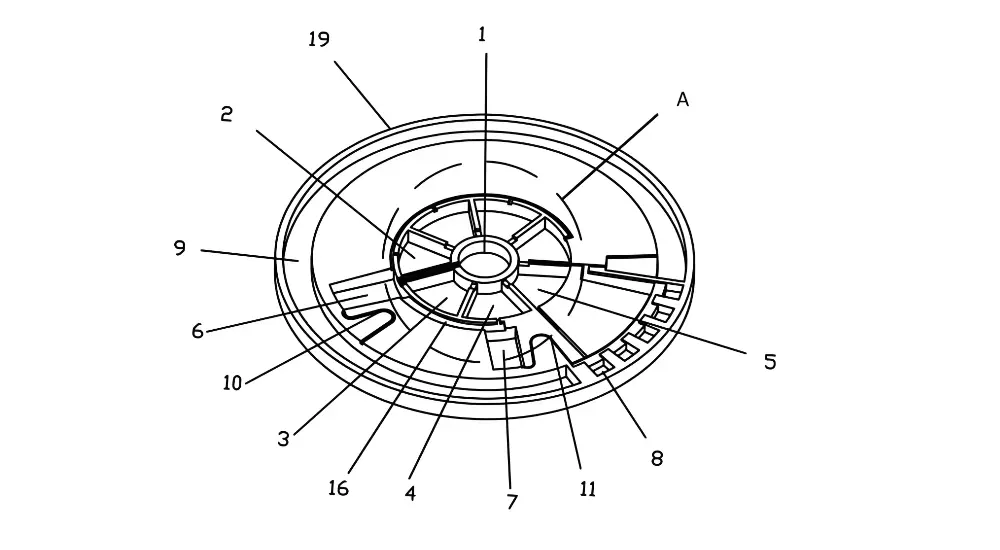
How a Centrifugal Chemistry Analyzer Works
When a disc is loaded into an analyzer, the instrument rotates it at controlled speeds to drive fluid movement. At low rotation speeds (typically 100–600 rpm), surface tension holds fluids in their loading chambers. At higher speeds (2,000–4,000 rpm), centrifugal force overcomes surface tension and drives precise volumes of reagent and patient sample outward into reaction cuvettes positioned at the disc periphery.
The photometric measurement step occurs while the disc spins at a stable speed. Light from a spectrometer passes through the optical windows of each cuvette radially; the absorbed wavelengths indicate the concentration of the target analyte. The entire process — from sample loading to result — typically takes 2–10 minutes. [1]
Types of Reagent Discs by Detection Method
Colorimetric / photometric discs: The most common type. Reagents produce a colour change proportional to analyte concentration, measured by absorbance at specific wavelengths. The optical windows in these discs must be flat to < 0.05 µm Ra and free from birefringence to prevent measurement error.
Fluorescence-based discs: Used for immunoassays and nucleic acid detection. These require materials with near-zero autofluorescence — cyclic olefin copolymer (COC) or cyclic olefin polymer (COP) rather than polycarbonate — because even small background fluorescence from the disc material interferes with the signal from the labelled analyte.
Electrochemical discs: Used for ion-selective measurements (electrolytes: sodium, potassium, chloride). Contain integrated reference and sensing electrode structures rather than optical windows. Material requirements prioritise chemical compatibility with the electrolyte reference solutions.
Where Chemistry Reagent Discs Are Used
Benchtop clinical chemistry analyzers in hospital laboratories use high-throughput reagent disc platforms for metabolic panels, liver function tests, lipid panels, and therapeutic drug monitoring. POCT instruments in emergency departments, outpatient clinics, and remote care settings use simpler single-patient disc formats that deliver results in 2–5 minutes.
Veterinary clinics deploy disc-based analyzers as a direct equivalent to human POCT platforms — the same centrifugal analysis principle is applied to blood chemistry panels in dogs, cats, and large animals. The disc designs are species-specific in their analyte panel configuration but share the same manufacturing requirements.
Why Injection Molding Is the Manufacturing Method for Reagent Discs?
Three manufacturing realities make injection molding the only practical route to commercial-scale reagent disc production: the geometrical complexity of microfluidic features, the optical quality requirements of measurement cuvettes, and the volume economics of disposable diagnostics.
Microfluidic Precision: Tolerances That Define Test Accuracy
Microfluidic channels in reagent discs are typically 50–500 µm wide and 50–200 µm deep. These dimensions control flow rates, mixing ratios, and the timing of reagent release under centrifugal conditions. A dimensional deviation of ±5 µm in channel width changes the hydraulic resistance by approximately 20%, altering the flow timing of the assay.
Injection molding achieves channel dimensions to ±0.01 mm through precision mold tooling (slow-wire EDM machining of cavity features to ±0.005 mm), controlled melt temperatures, and validated injection profiles that ensure complete feature replication without short shots or sink.
No other mass-production process matches this combination of feature resolution, dimensional consistency, and production rate. Hot embossing and soft lithography are used for laboratory prototyping, but neither scales to the production volumes diagnostic platforms require nor achieves the part-to-part consistency that validated diagnostic devices demand.
Structural Complexity in a Single Moulded Component
Modern reagent discs incorporate multiple functional structures within a single injection-moulded body: concentric arrays of reagent wells, radial transfer channels connecting inner loading chambers to outer reaction cuvettes, venting channels to allow air displacement during centrifugal filling, and optical windows positioned at precise radial locations for spectrophotometric measurement.
Producing all of these features as a single moulded component — rather than assembling them from separate machined parts — is only achievable through injection moulding with purpose-designed, high-precision multi-cavity tooling.
The alternative approach of machining individual disc components and assembling them would multiply the number of dimensional stack-up errors, increase the risk of particle generation during assembly, and make high-volume production economically non-viable.
Optical Quality Requirements
The measurement cuvettes in a reagent disc serve as optical cells through which the analyser’s photometer measures light absorbance. The optical path length — typically 1.0–6.0 mm — must be accurate and reproducible across every disc in a production lot.
The inner surfaces of the optical windows must be polished to SPI A-1 specification (Ra < 0.025 µm) to prevent light scatter. Mold steel for optical features is typically S136 stainless or H13 hardened to HRC 48–52, polished to mirror quality, and inspected for surface defects before production begins.
Volume Economics of Disposable Diagnostics
A single POCT analyzer platform can consume 5–20 million disc units annually across its installed base. Laboratory benchtop analyzers using multi-use disc formats still require several hundred thousand discs per year at major hospital networks. These volumes demand a manufacturing process whose cost per part decreases sharply with volume — exactly the characteristic that defines injection moulding economics.
Once a high-quality multi-cavity mold is qualified and validated, the marginal cost per disc is dominated by material cost. Labour and machine time per part are negligible at production scale. This cost structure is what makes high-performance diagnostics accessible at point-of-care settings globally.

Material Selection for Reagent Disc Injection Molding
Material selection is the first decision in reagent disc design — and the most consequential for assay performance. The chosen polymer must be optically compatible with the detection method, chemically inert to the reagent chemistries used in the assay, processable to the tight tolerances the disc geometry requires, and compliant with the biocompatibility framework for in vitro diagnostic devices. [2]
There is no universal material for reagent discs. The optimal choice depends on whether the assay uses photometric, fluorescence, or electrochemical detection; the reagent solvents and buffers involved; the thermal cycling requirements of the assay; and the target cost point of the disposable.
| Material | Form | Key Properties for Reagent Disc Applications | Typical Applications |
| Polycarbonate (PC) | Rigid, transparent | Impact-resistant, optically clear in the visible spectrum, withstands PCR thermal cycling (up to ~135°C). Small amounts of autofluorescence in fluorescence detection applications. | PCR reaction chambers, multi-cuvette rotors, centrifugal analyzer disc bodies, structural components requiring thermal cycling stability |
| Cyclic Olefin Copolymer (COC) | Rigid, transparent | Near-zero autofluorescence, excellent UV transparency, extremely low moisture absorption, broad chemical resistance to polar solvents. Premium optical properties for fluorescence-based assays. | Fluorescence detection discs, immunoassay cartridges, optical windows requiring UV transmission, high-sensitivity assay platforms |
| Cyclic Olefin Polymer (COP) | Rigid, transparent | Near-identical properties to COC with slightly better moisture barrier. Excellent birefringence control. Compatible with most organic solvents used in diagnostics. | High-sensitivity diagnostic discs, drug-compatible cartridges, applications requiring the lowest possible non-specific protein binding |
| Polystyrene (PS) | Rigid, clear | Low cost, optically clear in the visible range, good surface energy for biomolecule attachment (useful for ELISA and immunoassay substrates). Limited chemical resistance. | Immunoassay substrate discs, ELISA-format diagnostic plates, cost-sensitive high-volume platforms |
| Polypropylene (PP) | Semi-rigid, opaque | Low extractables, excellent chemical resistance, very low non-specific protein binding, cost-effective. High flexibility limits optical clarity — typically used for non-optical components. | Reagent storage chambers, sealing layers, low-binding sample contact surfaces, lids and covers for disc assemblies |
| PMMA (Acrylic) | Rigid, transparent | High optical clarity across visible spectrum, lower cost than COP/COC, excellent light transmission. Poor solvent resistance; can crack under stress. | Optical detection windows, photometric measurement cuvettes, visible-light-based colorimetric assay platforms |
COC and COP have emerged as the materials of choice for new fluorescence-based diagnostic platform designs. Their near-zero autofluorescence and excellent UV transparency open detection windows below 320 nm that polycarbonate cannot access. Their extremely low moisture absorption (< 0.01%) ensures that pre-loaded reagents stored within the disc remain stable over the product’s shelf life.
Polycarbonate remains dominant for photometric platforms because it provides the necessary optical clarity in the visible spectrum, sufficient heat resistance for PCR-temperature cycling (Tg ≈ 145°C), and an established regulatory track record in IVD device submissions.
Precision Manufacturing Requirements for Chemistry Reagent Discs
The requirements for injecting and inspecting reagent disc components differ substantially from general-purpose medical plastic production. The table below documents the specific manufacturing specifications that separate reagent disc production from standard medical component moulding.
| Requirement | Specification | Engineering Rationale |
| Microfluidic channel dimensions | ±0.01 mm or tighter | Channels as narrow as 50–200 µm control reagent flow without valves. Dimensional deviation of even 5 µm changes the flow resistance curve, altering assay timing and reaction volumes. High-speed mold filling must be balanced against feature replication fidelity. |
| Optical window flatness | Ra < 0.05 µm (mirror polish, SPI A-1) | Photometric and fluorescence measurements require windows with surface roughness that does not scatter light into the detector. Mold steel must be polished to mirror quality; any ejector pin marks or gate vestiges on the optical face are rejection criteria. |
| Reagent well volume accuracy | ±1–5% of nominal volume | In centrifugal analyzer rotors, each cuvette must hold a precise volume of reagent. Volume variation changes the reagent-to-sample ratio, directly affecting the accuracy of the biochemical result. Well depth, diameter, and base flatness are all critical features. |
| Wall thickness (optical path) | 0.05–0.2 mm (thin-wall molding) | PCR and temperature-cycling applications require ultra-thin wall sections to achieve rapid heat transfer. Filling these sections requires high-flow materials and validated injection profiles — standard process parameters produce incomplete fill or weld lines. |
| Cleanliness / contamination | ISO 14644 Class 7 or 8 cleanroom | Particulate contamination within microfluidic channels causes flow blockages that produce false results. Biological contamination can inhibit enzyme-based assay reactions. All production, inspection, and packaging steps must occur in validated cleanroom environments. |
| Bonding / sealing accuracy | < 0.05 mm layer alignment | Most reagent disc designs require bonding a cover layer or film to seal the microfluidic channels. Misalignment of even 0.05 mm can partially obstruct a channel, create a leak path, or expose reagent wells. Thermal or ultrasonic bonding parameters must be validated. |
Cleanroom Production and Contamination Control
Particulate contamination is the most common cause of reagent disc field failures. A single fibre or particle lodged across a microfluidic channel — typically 50–200 µm wide — causes a complete blockage, stopping the assay and producing a no-result error or false reading. Most IVD disc manufacturers specify ISO 14644-1 Class 7 (ISO 7) or Class 8 production environments for all molding, inspection, and assembly operations. [3]
Contamination control in reagent disc production goes beyond cleanroom classification. Resin handling, mold maintenance, and packaging must all occur in controlled environments. Molds must be cleaned and inspected regularly — any mold release agent residue or polymer degradation product can contaminate the disc surface and interfere with reagent chemistry.
Process Validation and IQ/OQ/PQ
Reagent disc injection molding is a special process under ISO 13485 Clause 7.5.6 — the output cannot be fully verified by downstream dimensional inspection alone. Microfluidic channel cross-sections, optical window surface quality, and internal wall integrity all require validated process controls, not just end-of-line inspection.
IQ/OQ/PQ validation for reagent disc programmes documents the processing window (barrel temperature range, injection pressure range, cooling time range) within which Cpk ≥ 1.33 on all critical dimensions is maintained. For critical-to-quality features such as cuvette path length and channel width, Cpk ≥ 1.67 is the standard target in regulated IVD programmes.
→ See also: Fecision medical injection molding capabilities
Clinical and Point-of-Care Applications
The same precision moulding platform serves diagnostic applications across the full clinical pathway — from central laboratory analysis through emergency department POCT to remote community health settings.
Clinical Chemistry Panels
Metabolic panels, lipid panels, liver function tests, and renal function tests are the highest-volume applications for reagent disc platforms. A single comprehensive metabolic panel disc may contain 12–24 discrete cuvettes — each requiring precise reagent volume, correct optical path length, and hermetic sealing — all produced in a single moulding cycle.
Cardiac Biomarker Testing
Cardiac troponin, BNP, and CK-MB testing in emergency department settings requires results within 15–30 minutes of sample collection. Disc-based POCT platforms achieve this by eliminating sample transport and batch processing. The disc designs for cardiac testing typically use fluorescence immunoassay detection, driving material selection toward COC or COP for autofluorescence-free performance.
Veterinary and Remote Diagnostics
Portable disc analysers enable blood chemistry testing at the point of care in veterinary practices, field hospitals, and remote health outposts where laboratory infrastructure is unavailable. The same injection-moulded disc format used in hospital POCT systems is adapted for veterinary species-specific panels — the manufacturing requirements are identical, but the reagent configurations differ.
Drug of Abuse and Therapeutic Drug Monitoring
Disc-based platforms for therapeutic drug monitoring (immunosuppressants, antiepileptics) and rapid toxicology screening use antibody-based immunoassay chemistries loaded into the disc wells at manufacturing. The material compatibility requirement is stringent — the disc polymer must not leach any compound that could inhibit the antibody-antigen reaction or bind non-specifically to the assay antibodies.

Emerging Developments in Reagent Disc Manufacturing
The design and production of chemistry reagent discs continue to evolve alongside diagnostic technology requirements. Three specific developments are reshaping what injection moulding for diagnostic discs can achieve.
In-Mold Labeling and Integrated RFID
Embedded identification features — including in-mould label inserts containing 2D barcodes or RFID chips — allow analyzers to automatically read disc type, lot number, reagent calibration data, and expiry status without operator input. This removes transcription errors and enables automated quality control within the diagnostic workflow.
Injection moulding is uniquely suited to integrating these features because the insert or label can be placed in the mould cavity before injection — the same process used for metal insert moulding in structural applications. The plastic flows around the insert and seals it permanently without adhesive.
Biodegradable Polymer Discs
Regulatory and sustainability pressures are creating interest in biodegradable materials for single-use diagnostic consumables. Polylactic acid (PLA) and polyhydroxyalkanoates (PHA) are being evaluated for diagnostic disc applications where service temperatures and reagent chemistries are compatible with their properties.
The transition from conventional thermoplastics to biodegradable grades requires full re-qualification of tooling, process parameters, and material biocompatibility.
Microcellular Foaming and Structural Integration
Microcellular foaming (Mucell process) creates a uniform micro-cellular structure within the disc body that reduces material use and weight without compromising the optical and fluidic precision of the functional features. The technique is being applied to the structural (non-optical, non-fluidic) regions of reagent discs to reduce material cost while maintaining performance where it matters.
Fecision — Injection Molding for Diagnostic Consumables
Fecision’s precision injection molding capability covers the full production requirements for diagnostic disc components — from high-cavitation production tooling through cleanroom molding and IQ/OQ/PQ validation. Our ISO 13485:2016-certified quality management system provides the regulatory documentation framework required for IVD device submissions.
- Dimensional precision: Slow-wire EDM tooling to ±0.005 mm on microfluidic cavity features. CMM inspection with Cpk ≥ 1.67 on critical dimensions.
- Optical quality: S136 stainless steel mould cavities polished to SPI A-1 (Ra < 0.025 µm) for photometric window surfaces.
- Cleanroom production: ISO 7 (Class 1,000) cleanroom moulding, assembly, and packaging. Particle monitoring per shift.
- Material capability: PC, COC, COP, PS, PP, PMMA — all medical-grade, with CoA and biocompatibility documentation per lot.
- Validation support: IQ/OQ/PQ protocols, dimensional reports, and traceability records formatted for FDA 510(k) and CE Technical File inclusion.
Contact us to discuss reagent disc or diagnostic consumable programs!
Frequently Asked Questions
What plastic is used to make chemistry reagent discs?
The most common materials are polycarbonate (PC), cyclic olefin copolymer (COC), and cyclic olefin polymer (COP).
- PC is used for photometric (colorimetric) detection platforms because it is optically clear, cost-effective, and withstands PCR-temperature thermal cycling.
- COC and COP are used for fluorescence-based platforms because they have near-zero autofluorescence — a critical requirement when detecting labelled targets at low concentrations.
- Polypropylene and polystyrene are used for specific components within the disc where optical clarity is not required.
Can injection-moulded reagent discs be made from biodegradable plastic?
Biodegradable polymers such as PLA (polylactic acid) are being evaluated for diagnostic disc applications, but none have yet achieved wide commercial adoption in regulated IVD products.
The challenges are material property consistency (PLA’s properties vary with crystallinity and molecular weight), limited chemical resistance to some assay buffers, and the regulatory requirement to fully re-qualify any material change under ISO 13485.
References & Sources
Accessed April 2026.
[1] U.S. Patents 4,226,531; 4,314,970; 4,678,331; 5,266,268 — Centrifugal multicuvette rotor design for clinical chemistry analyzers (Abbott, American Monitor). https://image-ppubs.uspto.gov/dirsearch-public/print/downloadPdf/4226531
[2] Parallel Fluidics / Microfluidics Innovation Center. ‘Material Selection for Microfluidic Devices.’ https://www.parallelfluidics.com/resources/knowledge-base/material-selection-for-microfluidic-devices
[3] ISO 13485:2016 — Medical devices: Quality management systems. Clause 7.5 (Production and service provision) and Clause 7.5.6 (Validation of processes for production). https://www.iso.org/standard/59752.html



