| An implantable medical device is a device placed inside or on the surface of the human body, either surgically or through a clinical procedure, to restore function, deliver therapy, monitor physiology, or provide structural support. The FDA classifies most implantable devices as Class III — the highest-risk category — requiring Premarket Approval (PMA) and independent clinical evidence of safety and effectiveness. |
Implantable devices range from a titanium orthopaedic screw costing a few hundred dollars to a fully programmable neurostimulator managing chronic pain. What they share is a demanding regulatory and engineering framework — because failures inside the human body cannot be easily corrected and the consequences can be catastrophic.
This guide covers the main device types, how materials are selected and evaluated for biocompatibility, manufacturing processes and process validation, and the regulatory pathway manufacturers must navigate from concept to market approval.
What Are Implantable Medical Devices?
An implantable device is introduced into the body — typically during surgery — to perform a function that the body cannot perform independently or to correct a condition that cannot be managed with external devices alone. The implant may remain permanently, be absorbed over time, or be intended for retrieval after a defined service period.
The defining characteristic is tissue contact duration. ISO 10993-1:2025 defines implantable devices as those with permanent contact (>30 days) with bone, dentin, or tissue — the contact duration category that triggers the most comprehensive biological safety evaluation requirements. [1]
Active vs. Passive Implants
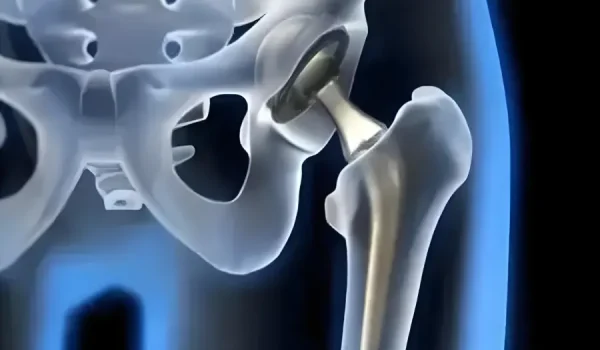
Passive implants provide structural support, containment, or mechanical function without onboard power. Hip replacement components, spinal fusion cages, vascular stents, and IUDs are passive implants. Their design priorities are mechanical strength, wear resistance, long-term chemical stability, and osseointegration or tissue compatibility at the interface.
Active implantable medical devices (AIMDs) rely on stored energy or external power to sense signals, deliver electrical stimulation, or pump fluids. Pacemakers, implantable cardioverter defibrillators (ICDs), left ventricular assist devices (LVADs), and spinal cord stimulators are AIMDs. Their design must address battery longevity, telemetry reliability, hermetic electronics packaging, and thermal safety.
FDA Device Classification: Class I, II, and III
The FDA classifies medical devices into three risk categories. Classification determines the regulatory pathway, clinical evidence requirements, and post-market obligations.
| Class I | Class II | Class III | |
| Risk level | Low risk | Moderate-to-high risk | High risk — life-sustaining or life-supporting |
| Example devices | Dental floss, elastic bandages, non-powered surgical instruments | Powered wheelchairs, infusion pumps, surgical drapes | Pacemakers, ICDs, LVADs, spinal cord stimulators, deep brain stimulators |
| FDA review pathway | General Controls only — some are exempt from premarket review | 510(k) Premarket Notification (substantial equivalence to a predicate) | Premarket Approval (PMA) — independent demonstration of safety and effectiveness |
| Clinical data required | Not required in most cases | Typically not required if strong predicate exists | Required — IDE (Investigational Device Exemption) clinical trial data in most cases |
| QMS requirement | 21 CFR Part 820 (QMSR, effective Feb 2026) | 21 CFR Part 820 (QMSR) | 21 CFR Part 820 (QMSR) + ISO 13485 preferred by FDA for PMA submissions |
| Post-market duties | MDR reporting, corrections, removals | MDR reporting, corrections, removals, periodic reports | MDR reporting, PMA supplements for design changes, post-approval studies |
Most implantable devices — pacemakers, spinal implants, cochlear systems, neurostimulators — are classified as Class III. This means the manufacturer must submit a Premarket Approval (PMA) application supported by clinical trial data, typically gathered under an FDA-approved Investigational Device Exemption (IDE). [2]
Types of Implantable Medical Devices
Implants cover a wide range of anatomical locations, clinical functions, and engineering architectures. The categories below represent the major device families from a manufacturing and regulatory standpoint.
Cardiac Rhythm Management Devices
Pacemakers deliver timed electrical impulses through leads placed in heart tissue to maintain cardiac rhythm and rate. Long-term pacemakers — designed for service lives of 7–10+ years — require durable lithium batteries, secure lead fixation, and wireless telemetry for remote follow-up. Lead fracture and insulation breach are the primary long-term failure modes.
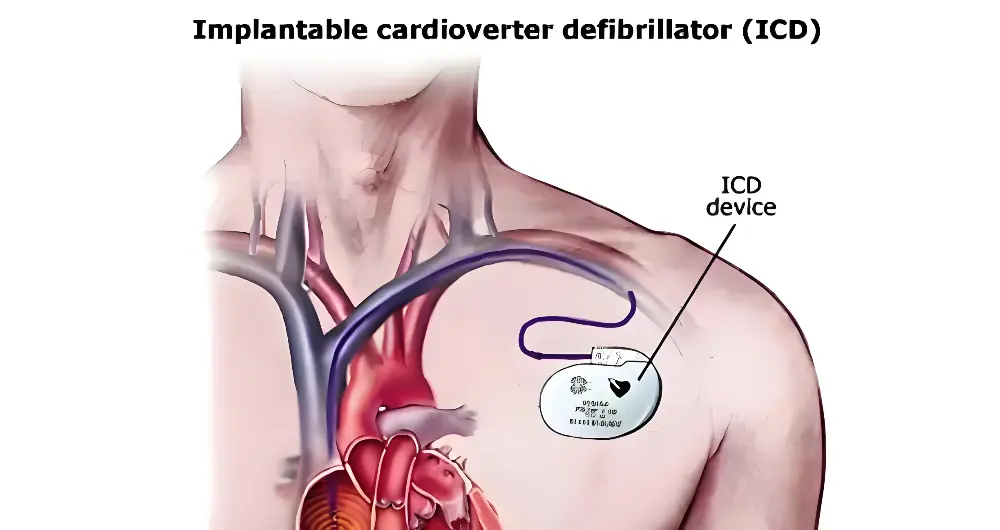
Implantable cardioverter defibrillators (ICDs) incorporate both pacing capability and the ability to detect and terminate life-threatening arrhythmias (ventricular tachycardia, ventricular fibrillation) with a high-voltage shock. The device housing (commonly titanium) must provide electromagnetic shielding, hermetic sealing against body fluids, and shock-wave durability.
Left ventricular assist devices (LVADs) are surgically implanted pumps that move blood from the left ventricle to the aorta, partially or fully replacing left ventricular pump function. LVADs serve as either a bridge to transplant or — increasingly — as destination therapy for patients who are not transplant candidates. They require portable external controllers, redundant alarm systems, and robust driveline sealing where the device exits the body.
Musculoskeletal and Orthopaedic Implants
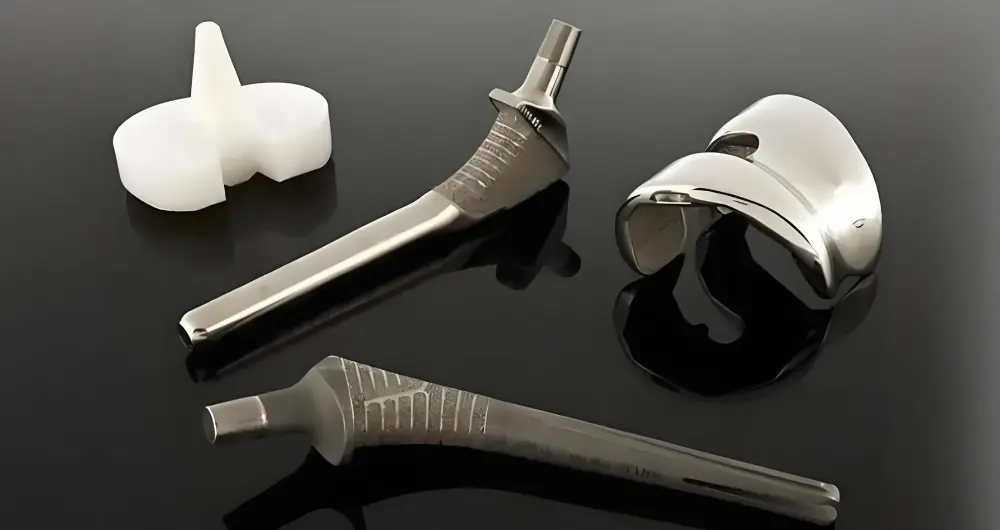
Total joint replacements — hips, knees, shoulders, ankles — are among the highest-volume implantable devices manufactured globally. They couple bearing surfaces with fixation components, each demanding different material properties from the same assembly.
The femoral head of a hip replacement, for example, requires extremely high hardness and surface smoothness on the articulating face (ceramic or CoCr, surface roughness Ra < 0.02 µm) while the stem requires bone-bonding capability or cementless fixation through porous coating geometry. Spinal implants — cages, rods, pedicle screws — prioritise load distribution and osseointegration without creating artefact in CT or MRI imaging.
Neurological and Sensory Implants
Cochlear implants convert sound to electrical signals delivered via an electrode array positioned in the cochlea. The external processor communicates wirelessly with a surgically implanted receiver-stimulator. Materials requirements include MRI compatibility, long-term titanium hermetic housing integrity, and platinum electrode array corrosion resistance.
Deep brain stimulators (DBS) and spinal cord stimulators deliver targeted electrical stimulation to modulate neural activity in conditions including Parkinson’s disease, essential tremor, and chronic pain. Lead placement precision is critical — electrode position relative to target anatomy determines therapeutic efficacy and side-effect profile.
Intraocular lenses (IOLs) replace the natural lens after cataract extraction. Materials are hydrophilic or hydrophobic acrylic, with exceptional optical clarity, UV filtering, and long-term refractive stability requirements. IOLs are among the highest-volume implantable devices by unit count globally.
Vascular and Fluid Management Implants
Stents — coronary, peripheral, and biliary — provide scaffolding to maintain vessel or duct patency. Drug-eluting stents (DES) incorporate a polymer coating and therapeutic agent that releases over weeks to months to inhibit restenosis. The combination of structural, drug-delivery, and biocompatibility requirements makes DES one of the most demanding regulatory evaluation programs in Class III devices.
Implanted vascular access ports — subcutaneous chambers connected to central venous catheters — provide long-term chemotherapy or parenteral nutrition access without repeated venepuncture. They require infection resistance, needle-septum durability (rated to 1,000–2,000 needle insertions), and heparin lock compatibility.
Drug Delivery Implants
Implantable drug delivery systems achieve sustained therapeutic levels that oral or injected drugs cannot maintain reliably. Osmotic pumps deliver nanolitre-per-hour flow rates of biologics or analgesics. Subcutaneous implants — such as hormonal contraceptive rods — release controlled amounts of active pharmaceutical ingredient over years.
Materials for Implantable Medical Devices
Material selection is the most consequential design decision in any implantable device program. The chosen material must deliver the required mechanical performance, maintain its properties over the device’s service life inside the body, and satisfy the biological safety evaluation required under ISO 10993.
Material Selection Overview
| Material | Class | Key Properties | Typical Implant Applications |
| Titanium (Ti-6Al-4V) | Metal | Tensile strength 950 MPa, excellent fatigue life (>10⁷ cycles), MRI-compatible, low density (4.5 g/cm³) | Orthopaedic implants, pacemaker housings, spinal fusion cages, dental root analogs |
| Cobalt-Chromium (CoCrMo) | Metal | Higher hardness than titanium, superior wear resistance on articulating surfaces, proven 30+ year clinical history | Total hip and knee replacement bearing surfaces, cardiac valve components |
| 316L Stainless Steel | Metal | Good strength and corrosion resistance, lower cost than Ti or CoCr, less MRI-compatible | Temporary fixation hardware, surgical instruments, short-term structural implants |
| PEEK | Polymer | Modulus 3.6 GPa ≈ cortical bone (3–30 GPa), radiolucent, autoclave-stable, chemically inert | Spinal interbody fusion cages, trauma fixation plates, implantable electronic enclosures |
| UHMWPE | Polymer | Exceptional wear resistance, low friction coefficient, impact resistant, FDA 510(k) cleared for articular use | Acetabular cup liners, tibial trays, total disc replacement nucleus |
| Silicone (implant grade) | Elastomer | Soft, flexible, chemically inert, biocompatible, available in Class VI and ISO 10993 tested grades | Cochlear implant components, breast implants, shunt valves, pacemaker lead insulation |
| ePTFE (Gore-Tex) | Polymer | Microporous structure promotes tissue ingrowth, blood-contacting biocompatibility, kink-resistant | Vascular grafts, hernia mesh, soft-tissue reconstruction patches |
| Alumina / Zirconia | Ceramic | Hardness 1,500–2,000 HV, near-zero wear in vivo, no metal ion release, radiopaque | Femoral head components, dental abutments, bearing surfaces where metal sensitivity is a concern |
| Hydroxyapatite (HA) | Bioceramic | Chemical composition matches natural bone mineral, stimulates osseointegration, can be coated on metal | Orthopaedic implant coatings, dental implant surfaces, bone void filler |
| Bioabsorbable PLA / PLGA | Polymer | Degrades predictably over months to years, eliminates secondary removal surgery, supports bone healing | Fixation screws for small fractures, drug-eluting stents, temporary structural scaffolds |
Material pre-qualification for implantable use follows a hierarchy.
- USP Class VI testing (three in vivo reactivity tests) establishes a baseline.
- ISO 10993 evaluation adds cytotoxicity, sensitisation, implantation, haemocompatibility, genotoxicity, chronic toxicity, and carcinogenicity testing depending on contact type and duration.
- Long-term implants (>30 days) require the full biological evaluation battery.
ISO 10993-1:2025 — Key Updates for Implantable Devices
ISO 10993-1:2025 (the sixth edition, published November 2025) introduced several changes relevant to implantable device manufacturers:
- Expanded genotoxicity requirements: All devices with prolonged (>24 hours to 30 days) or long-term (>30 days) contact now require genotoxicity evaluation, regardless of contact site. This is a broadening from the 2018 edition.
- ‘Foreseeable misuse’ assessment: Manufacturers must now evaluate biological risks associated with reasonably foreseeable misuse, not just intended use. If misuse could extend contact duration or change contact site, the biological evaluation must address that scenario.
- Restructured exposure categories: The four separate evaluation matrices (replacing the single Table A.1 in the 2018 edition) provide more specific guidance by contact type — relevant for complex implants with multiple material interfaces.
- Lifecycle-based approach: The standard now aligns explicitly with ISO 14971 risk management throughout the biological evaluation process, not only at the test selection stage. [1]
Devices already on the market do not automatically require new testing under ISO 10993-1:2025. However, manufacturers must review existing Biological Evaluation Plans (BEPs) and document that previous evaluations remain valid. If gaps are identified — for example, absent genotoxicity data for a long-term contact device — additional testing is required.
Surface Treatments and Coatings
Surface condition often matters more than bulk material composition for biological performance. The key surface treatments for implantable devices include:
- Titanium anodisation and grit-blasting: Creates a rough, oxide-rich surface that promotes osteoblast attachment and bone ingrowth. Standard on cementless orthopaedic implants.
- Hydroxyapatite (HA) coating: Plasma-sprayed HA on implant surfaces mimics natural bone mineral, accelerating osseointegration. Coating adhesion strength and dissolution rate must be validated.
- Passivation (stainless steel, CoCr): Acid passivation removes free iron from the surface and thickens the passive oxide layer, reducing corrosion ion release in the body. Required per ASTM F86 for stainless implants.
- Drug elution coatings: Polymer matrices loaded with antiproliferative agents (paclitaxel, sirolimus) on coronary stents require validation of drug release kinetics, coating adhesion, and degradation products.

Manufacturing Implantable Medical Devices
Manufacturing implantable devices is not simply high-precision fabrication — it is a fully documented, validated process where every production parameter is controlled and every deviation generates a formal record.
Precision CNC Machining
CNC machining is the primary fabrication route for titanium, CoCr, and stainless steel implant components. It achieves tolerances of ±0.005mm or tighter on critical features — bone-contact surfaces, locking screw interfaces, and bearing geometry. Multi-axis mill-turn centres produce complex spinal implant geometries in a single setup, eliminating the datum-shift errors introduced by refixturing.
Hard milling of ceramic implant components (alumina, zirconia) requires diamond-tipped tooling and controlled feed rates to avoid subsurface cracking that would not be detectable by surface inspection. Post-machining sintering and polishing bring ceramic femoral heads to surface roughness Ra < 0.02 µm — a requirement for acceptable in vivo wear performance.
→ See: Fecision precision CNC machining capabilities
Injection Molding for Polymer Implant Components
Medical-grade injection molding produces PEEK spinal cages, UHMWPE bearing liners, and silicone elastomer components. The process parameters — barrel temperature, injection pressure, cooling rate — must be qualified and controlled, not just set. Variation in crystallinity (for semi-crystalline PEEK) directly affects mechanical stiffness and fatigue performance.
Cleanroom injection molding (ISO 7 or ISO 8 environment) is standard for implant-grade polymer components. Material lot traceability — from resin batch CoA through production records to finished part — is a mandatory documentation requirement under ISO 13485.
→ See: Fecision medical injection molding
Additive Manufacturing for Patient-Matched Implants
Laser powder bed fusion (LPBF) and electron beam melting (EBM) produce titanium orthopaedic implants with patient-matched geometry and designed porosity for bone ingrowth. Internal lattice structures — impossible with subtractive machining — allow modulus-matching to cortical bone, reducing stress shielding that causes long-term bone resorption around conventional dense-metal implants.
Regulatory expectations for additive-manufactured implants are more demanding than for conventionally manufactured equivalents. Consistent powder properties, build orientation, and laser parameters must all be validated and communicated to the FDA. Degradation testing and sterility validation strategies for AM implants require case-by-case FDA consultation. [3]
Electronics Integration for Active Implants
Active implants require hermetically sealed electronics packages. The hermetic feedthrough — a glass-to-metal or ceramic-to-metal sealed electrical conductor — is the most critical manufacturing element. It must maintain hermeticity to a helium leak rate of less than 1×10⁻⁸ atm·cc/s through 10+ years of in vivo service, thermal cycling, and mechanical vibration from the patient’s activity.
Battery selection and packaging for AIMDs must balance energy density, cycle life, and safety under abuse conditions. Lithium-iodide batteries (non-rechargeable pacemakers) and lithium-ion systems (rechargeable neurostimulators) each have distinct hermeticity, thermal, and regulatory requirements.
Sterilisation Selection and Validation
Sterilisation method selection must be made before materials are finalised — not after. The table below maps common methods to compatible and incompatible implant materials.
| Method | Compatible Materials | Incompatible / Caution | Notes for Implantable Devices |
| Ethylene Oxide (EtO) | Most polymers, electronics | Metals, glass | Low-temperature process — compatible with heat-sensitive plastics. Requires 12–24 hr aeration to remove EtO residuals. Validated per ISO 11135. |
| Gamma Irradiation | Metals, ceramics, PE, PP | PVC (discolours), PTFE (degrades at high dose) | 25–50 kGy typical dose. Penetrates dense assemblies. Radiation-stabilised grades available for sensitive polymers. Validated per ISO 11137. |
| E-Beam | PP, PE, metals | Dense assemblies (lower penetration) | Similar to gamma but faster. Less penetrating — preferred for thin-walled single-use devices. Cannot sterilise thick or complex implant assemblies effectively. |
| Steam Autoclave (121–134°C) | PEEK, PPSU, metals, glass | PC (limited cycles), most elastomers | Reusable surgical instruments and implants designed for re-sterilisation. PEEK and PPSU handle repeated autoclave cycles without degradation. Cannot be used for electronics. |
| Dry Heat | Metals, glass, ceramics | All polymers | 160–180°C for 1–2 hours. Used for heat-stable metal components. Does not penetrate complex assemblies — only surface sterility is achieved. |
All sterilisation processes for implantable devices must demonstrate a Sterility Assurance Level (SAL) of 10⁻⁶ — meaning fewer than one non-sterile device in one million — validated per ISO 11135 (EtO), ISO 11137 (radiation), or ISO 17665 (steam). Packaging must maintain sterility through the validated shelf life, typically 5 years for most single-use implants.
Manufacturing Challenges Specific to Implantable Devices
Several challenges in implantable device manufacturing have no direct parallel in general precision manufacturing. Understanding them is essential for design teams planning a development programme.
Long-Term Biocompatibility of Multi-Material Assemblies
A single implantable device frequently combines metals, polymers, ceramics, and coatings in intimate contact. Galvanic corrosion between dissimilar metals, coating delamination releasing particles, or polymer degradation releasing leachables can all produce biological responses that were not present at initial device implantation.
The FDA explicitly notes that if a metal stent has a polymer coating that may separate over time, testing of the complete device may not fully reflect longer-term clinical performance — and biocompatibility evaluation of the stent with and without the coating may both be needed. [3]
Miniaturisation Limits for Active Implants
Smaller active implants mean shorter battery life, reduced telemetry power, and narrower tolerances on hermetic feedthroughs. Every element of an AIMD — battery chemistry, enclosure geometry, electrode design, telemetry frequency — involves trade-offs between size, performance, and longevity.
Thermal management is an underappreciated constraint. FDA expects characterisation of thermal output at maximum operating conditions.
Cybersecurity for Connected Active Implants
Wireless connectivity in modern pacemakers, neurostimulators, and insulin pumps introduces attack surfaces that did not exist in earlier generations of these devices. The FDA’s 2023 cybersecurity guidance for medical devices requires manufacturers to address cybersecurity throughout the device lifecycle — from threat modelling during design through software updates in the field.
Conclusion
Implantable medical devices occupy the most demanding space in the medical device industry — highest regulatory class, most extensive biocompatibility evaluation, most rigorous manufacturing validation, and post-market obligations that continue for the service life of every implanted unit.
The engineering decisions made at the design phase — material selection, manufacturing process, sterilisation method, hermeticity approach — establish the regulatory and clinical outcome for the device.
Starting with a manufacturing partner who understands both the technical requirements and the regulatory documentation framework — and can validate their process to the Cpk standards that Class III submissions require — is the most effective risk mitigation available in the early phase of an implant programme.
References & Authoritative Sources
[1] ISO 10993-1:2025. Biological evaluation of medical devices. https://www.iso.org/standard/10993-1
[2] U.S. Food and Drug Administration. ‘Overview of Device Regulation. https://www.fda.gov/medical-devices/overview-device-regulation/classify-your-medical-device
[3] U.S. Food and Drug Administration. ‘Use of International Standard ISO 10993-1, Biological Evaluation of Medical Device’. https://www.fda.gov/media/142959/download