LSR Injection Molding Services - Medical Grade Silicone Parts
Start Your Project Now
ISO 9001:2015
ISO 14001:2015
ISO 45001: 2018
ISO 13485: 2016
AS9100 Rev D
QC080000
High-quality LSR Injection Molded Parts
Our LSR Injection Molding Services
Fecision's LSR injection molding cell operates within a Class ISO 7 / 100,000 cleanroom, producing million+ precision silicone components per month for medical device OEMs, electronics manufacturers, and industrial customers

LSR Injection Molding Definition
LSR injection molding injects a two-part liquid silicone compound (base + catalyst in 1:1 ratio) into a heated mold where it vulcanizes into a permanent, cross-linked elastomer. Unlike thermoplastics that can be remelted, LSR's thermoset nature ensures dimensional stability and performance retention across extreme temperatures (-60°C to 230°C).
Fecision's Service
At Fecision, our ISO 13485:2016 quality management system governs every medical program — from incoming material certification through production, first-article CMM inspection (Cpk ≥1.67 on critical features), and full batch traceability. PPAP submission support is available for medical device customers requiring supplier qualification documentation.
What is Liquid Silicone Rubber (LSR)
Understanding LSR Injection Molding
Liquid Silicone Rubber (LSR) is a high-purity, versatile thermoset elastomer renowned for its exceptional performance characteristics. It is a two-part, platinum-cured silicone system (typically an A component and a B component mixed in a 1:1 ratio) that, once cured, offers a unique combination of properties making it an ideal choice for a vast array of demanding applications.
★ Outstanding thermal stability
★ Excellent chemical resistance
★ Inherent biocompatibility and hypoallergenic nature
★ Superior durability and tear strength
★ Electrical insulation properties
★ Can be easily pigmented for color-coding or aesthetic purpose
| Property | Typical Range | Standard |
| Hardness (Shore A) | 10 – 80 | ISO 7619-1 |
| Tensile Strength | 7 – 12 MPa | ISO 37 |
| Elongation at Break | 400% – 800% | ISO 37 |
| Tear Resistance | 17 – 47 kN/m | ISO 34-1 |
| Density | 1.11 – 1.20 g/cm³ | ISO 2781 |
| Temperature Range | -60°C to +230°C | – |
LSR Injection Molding Expertise
Fecision's LSR Injection Molding Capabilities

Advanced Equipment

Material Expertise

Precision Tooling

Value-Added Services
LSR Two-Shot Overmolding and Multi-Material Components
Fecision produces combination components where LSR is directly moulded onto thermoplastic substrates in a single production cell — eliminating secondary assembly and adhesive bonding steps.
Compatible substrate materials
PA6, PA66, PBT, PC, and other engineering plastics validated for LSR adhesion. Substrate surface activation (plasma treatment or Wacker Primer G790) is specified based on the material pairing and validated before production release.
Strength
Two-shot overmolded components eliminate: separate bonding operations · adhesive outgassing risk in cleanroom environments · delamination failure modes in service · BOM complexity and assembly labour cost.
Applications
Soft-seal medical connector housings where silicone seals onto rigid PC or PA bodies · Ergonomic surgical instrument handles with silicone grip overmoulded onto stainless steel shafts · Wearable medical device interfaces with LSR skin-contact layer on rigid thermoplastic backing · Audio and tactile button assemblies for medical devices and electronics.
The LSR Injection Molding Process
At Fecision, the LSR injection molding process is a highly precise and automated manufacturing method designed to produce complex, high-quality silicone parts with exceptional consistency and efficiency. Our process leverages advanced technology and stringent controls to ensure optimal results for every component.
Process Parameters Summary
Material storage: 20-25°C
Mixing ratio: 1:1 (Part A:Part B)
Mold temperature: 150-200°C
Injection pressure: 50-150 MPa
Curing time: 5-30 seconds
Cycle time: 30-90 seconds
Supplier Qualification Support
For components requiring pre-sterilization packaging qualification, Fecision provides cleanroom packaging validated per ISO 11607.
- Sterilization compatibility: Fecision medical LSR components have been validated for EtO, gamma irradiation, e-beam, and autoclave (134°C steam) sterilization methods.
- Packaging options: Sterile barrier pouches and trays validated for the customer’s specified sterilization method. Accelerated aging per ASTM F1980 and distribution simulation per ASTM D4169 are available as value-added services for programs requiring complete packaging validation documentation.
Cleanroom Packaging and Sterilization Support
Fecision is designed to pass your supplier qualification audit.
Our ISO 13485:2016 QMS documentation, equipment calibration records, process validation files, and environmental monitoring logs are maintained audit-ready at all times.
What we make available for supplier qualification:
• ISO 13485:2016 certificate and scope statement
• Quality Manual and relevant SOPs
• IQ/OQ/PQ reports for customer-specific processes (under NDA if required)
• Equipment calibration and preventive maintenance records
• Environmental monitoring data (cleanroom particle counts, temperature/humidity logs)
Fecision LSR Injection Molding Equipments
Our dedication to quality is validated by an extensive portfolio of prestigious certifications, demonstrating our adherence to the most rigorous global standards.
Design Considerations for LSR Injection Molding
Why It Matters for LSR
Injection prLSR fills thin sections readily — but uniform walls matter for cure consistency, not filling. Uneven walls produce uneven cure and part-to-part property variation.essure: 50-150 MPa
Why It Matters for LSR
LSR's flexibility allows more aggressive undercuts than thermoplastics. Standard thermoplastic draft rules are unnecessarily conservative for LSR materials.
Why It Matters for LSR
Undersized gates create shear heat even when barrel temperatures are correct. This is the most common cause of surface defects in LSR — not temperature control failure.
Why It Matters for LSR
Grade-specific shrinkage varies: harder grades (60–80 Shore A) shrink less than soft grades (20–40 Shore A). Post-cure also increases shrinkage by 0.3–0.5%.
Why It Matters for LSR
Inadequate venting produces burns (black marks at end of fill), short shots in micro-features, and weak weld lines behind inserts. Venting is often the last DFM check — it should be the first.
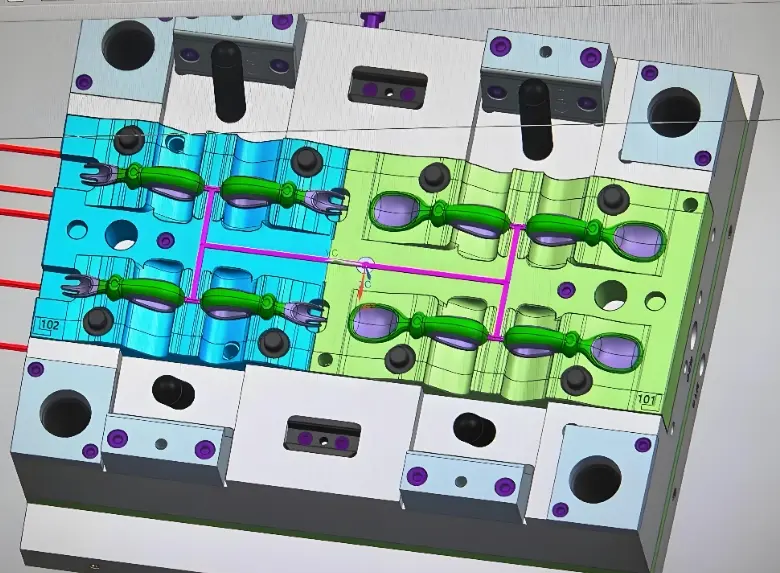
LSR vs Compression Molding: Which is Right for You?
Factor | LSR Injection Molding | HCR Injection Molding |
Material form | Two-part liquid (Part A + Part B) — fully automated metering | Solid gum-like material — requires manual or semi-auto feeding |
Viscosity | Low — fills thin walls, complex features, micro-geometries readily | High — requires more processing force; better for thick cross-section parts |
Tolerance | ±0.05mm achievable with cold runner tooling | ±0.15–0.3mm typical |
Cycle time | 30–90 seconds (fully automated) | 3–10 minutes (manual handling typical) |
Flash | Minimal with hardened, tight-shutoff tooling — often no trimming required | Requires deflashing — adds labour and cycle time |
Part complexity | High — intricate geometries, thin walls, internal features | Medium — simpler geometries preferred |
Automation | Fully automated from material delivery to part removal | Limited — manual material loading standard |
Cleanroom suitability | Excellent — closed material system, no operator contact with uncured material | Limited — open material handling increases contamination risk |
Best for (mechanical props) | High repeatability, tight tolerances, complex geometries | High tear strength for thick cross-section seals and specific implantable applications |
Volume economics | Most economical above ~15,000–30,000 pcs/year | More economical below ~15,000 pcs/year — lower tooling cost |
Advantages of LSR Injection Molding
Liquid Silicone Rubber (LSR) injection molding offers a multitude of advantages that make it a preferred manufacturing process for a wide range of critical components, particularly those requiring high precision, exceptional material performance, and suitability for demanding environments.
Complex Part Geometries & High Precision
The low viscosity of LSR allows it to flow easily into intricate mold cavities, enabling the production of highly complex part geometries, thin walls, and fine details with exceptional precision.Fecision's expertise in mold design and process control ensures that these complex designs are manufactured with tight tolerances and remarkable accuracy.
Excellent Material Properties
LSR's inherent durability and resistance to tearing and fatigue ensure long service life even under stress. LSR maintains its flexibility and mechanical properties across a broad temperature spectrum, from as low as -60°C to over 200°C, making it suitable for applications exposed to extreme thermal conditions. Furthermore, its excellent chemical inertness provides resistance to a wide array of chemicals, oils, and solvents. For medical and food-contact applications, the biocompatibility and hypoallergenic nature of specific LSR grades are paramount, ensuring safety and compliance.
Consistency & Repeatability
The LSR injection molding process delivers exceptional part-to-part consistency and repeatability.The stable nature of the LSR material and the precision of the molding process minimize variations, ensuring reliable performance of the final product.
Automation & Efficiency for Scalable Production
LSR's rapid curing cycle and the potential for automated demolding make the injection molding process highly efficient and suitable for large-scale production. This automation reduces cycle times, minimizes manual labor, and lowers per-part costs, especially in high-volume scenarios.
Inherent Cleanliness & Suitability for Sterile Applications
LSR is an inherently clean material, producing minimal outgassing and leachables. This characteristic, combined with the ability to mold parts in certified cleanroom environments makes LSR injection molding an ideal choice for medical devices, pharmaceutical components, and food-grade applications where sterility and purity are non-negotiable. The smooth, non-porous surface of LSR parts also resists bacterial growth and is easy to clean and sterilize.
Applications & Industries Served
Medical & Healthcare
Applications
Electronics
Applications
Industrial Components
Applications

Your Strategic Partner for LSR Injection Molding Success
The Fecision Advantage: Why Partner With Us?
- Certifications: ISO 9001:2015 · ISO 13485:2016 · AS9100 Rev D · GMP 21 CFR Part 820 compliant cleanroom · QC080000 · ISO 14001:2015 · ISO 45001:2018
- Quality metrics: Cpk ≥1.67 on critical-to-quality dimensions (ISO 13485 Clause 8.2.4). Full batch traceability from raw material CoA to finished part release. PPAP submission support included with medical programs.
- 18+ years of LSR injection molding experience across medical, electronics, and industrial programs. Our engineers have qualified LSR processes for Class I, II, and III medical devices — including products requiring IQ/OQ/PQ process validation under ISO 13485 Clause 7.5.6.
- We design and manufacture 500+ moulds annually, including LSR injection moulds, LSR compression moulds, and two-shot LSR/thermoplastic tools.
- Manufacturing: 10,000+ sqm facility. Class ISO 7 cleanroom for medical and regulated programs. 50+ injection moulding machines including LSR-dedicated cells with integrated cold runner systems and automated metering.
- LSR-specific equipment: All-electric LSR injection presses — no hydraulic oil contamination risk for medical programs. Automated material delivery from sealed drums to injection nozzle — closed system, zero operator contact with uncured material.
From DFM review to production release — a typical Fecision LSR medical program:
- Week 1–2: DFM review + material grade selection + tooling design review
- Week 3–8: Mould fabrication (CNC + EDM + precision grinding)
- Week 9–10: T0 trial + first-article CMM measurement
- Week 11–12: OQ parameter optimisation across validated range
- Week 13–15: PQ validation — 3 consecutive production runs, Cpk ≥1.67 confirmed
- Week 16+: Production release with full documentation package
[Timelines are indicative and depend on part complexity and regulatory program requirements. Contact us for a program-specific schedule.]

Validated Performance, Guaranteed Quality
Quality & Certifications








Recent Projects

Nipple

Respiratory mask parts

Bottom silicone pad

Earplugs

Silicone strap
Audio button

Liquid-proof stopper (dialyser)

Earphone seal
Let's Start!
Take the next step towards realizing your high-performance Liquid Silicone Rubber components.
LSR Injection Molding FAQs
Every Fecision medical LSR program includes: IQ/OQ/PQ protocol execution and summary reports · CMM first-article report (Cpk ≥1.67 on critical features) · Material CoA per production lot · Full batch traceability records from raw material to part release
For a standard medical LSR program: DFM review and approval (1–2 weeks) → mould fabrication (4–6 weeks) → T0 trial and first-article measurement (1–2 weeks). Total DFM-to-first-article: typically 8–12 weeks depending on part complexity. Contact our team for a program-specific timeline — complex geometries, high-cavitation tools, or special material requirements will extend this.
Yes. Fecision welcomes customer quality audits as part of the supplier qualification process. Our ISO 13485:2016 QMS documentation, process validation records, and equipment calibration logs are maintained audit-ready. Remote documentation reviews and on-site audits can be scheduled with 2 weeks' notice.
LSR injection molding becomes most economical above approximately 15,000–30,000 parts per year, where automated cycle times (30–90 seconds) and zero flash trimming significantly reduce per-part cost vs. compression moulding. For lower volumes or development programs, we offer bridge tooling options. Contact our team to discuss the right approach — we will recommend the most cost-effective route for your specific program, not the most expensive one.
You Might Also Be Interested In
Silicone Molding for Medical Devices: The Complete Manufacturing Guide
Learn how LSR injection molding works for medical devices. Covers material grades, DFM, sterilization, IQ/OQ/PQ validation, and cleanroom requirements.

Complete Guide to LSR Injection Molding for Medical Devices
LSR injection molding injects platinum-cured silicone at 100–1,000 bar into a 170–230°C mold, curing in 30 s–2 min. Complete guide: process, Wacker grades, FDA/ISO compliance.

LSR Injection Molding vs. HCR Injection Molding: A Complete Guide for Medical Use
LSR injection molding vs HCR: cycle time, tooling cost, medical grade specs and a clear decision framework. Engineering data from Fecision’s silicone molding team.
